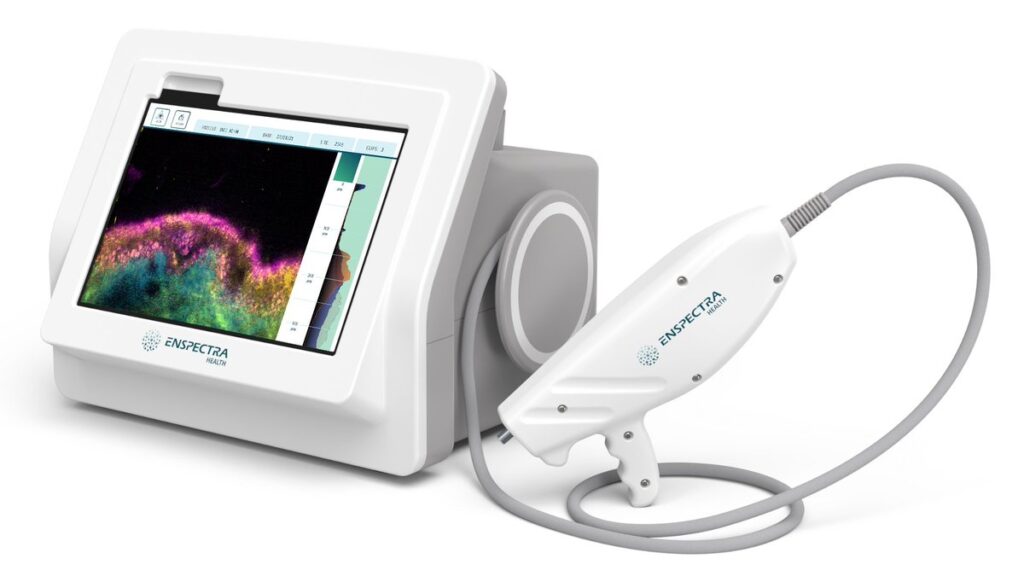
MOUNTAIN VIEW, Calif., June 5, 2024 /PRNewswire/ — Enspectra Health, a health tech company, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its next generation AI-powered VIO™ Skin Platform (VIO) for the evaluation of lesions suspicious of basal cell carcinoma (BCC) and squamous cell carcinoma (SCC) in select high risk populations. This novel platform integrates the FDA cleared VIO technology with VIO.ai NMSC, a CADx/CADt (Computer-Aided Diagnosis and Triage) software function that provides lesion classification to aid physicians in their clinical decision-making.
BCC and SCC are the most common forms of skin cancer and account for nearly all non-melanoma skin cancer (NMSC) diagnoses in the United States. While BCC and SCC generally pose lower mortality risk than melanoma, certain high-risk patients are much more likely to experience NMSC disease progression, increasing morbidity and mortality in these populations.
“When identified early, appropriate treatment can be delivered for non-melanoma skin cancer,” said Sarah T. Arron, M.D., Ph.D., board-certified dermatologist & Mohs surgeon and clinical advisor to Enspectra Health. “Early intervention aids in the prevention of cancer recurrence and spread.”