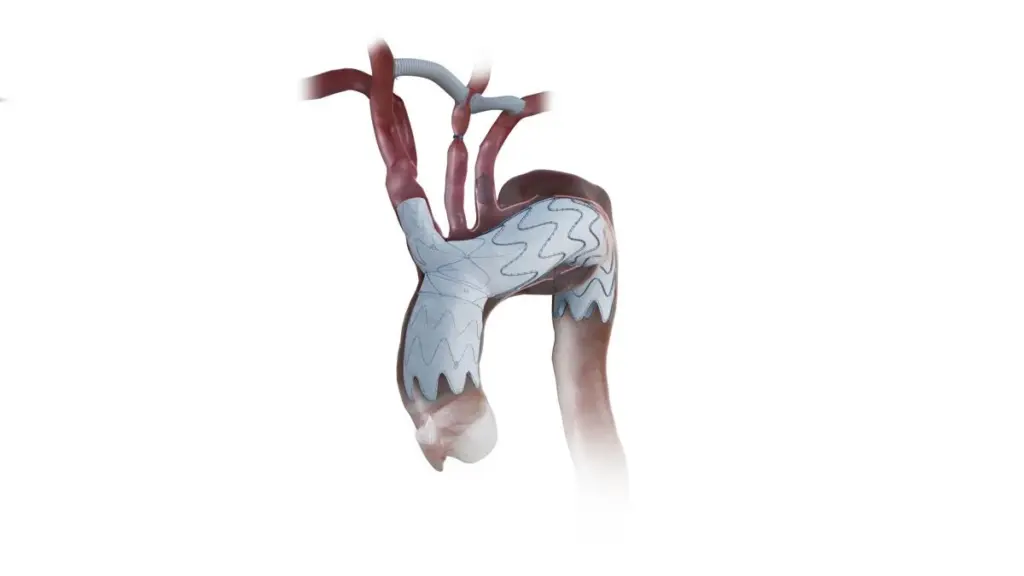
Israel’s Endospan has received US Food and Drug Administration (FDA) approval for the NEXUS aortic graft stent system, priming the company for the US commercialisation of the aortic arch dysfunction treatment.
Nexus is designed for treating high-risk surgical patients with aortic arch disease, which includes conditions such as chronic dissections of the aorta or aortic arch syndrome. The aorta, a cane-shaped vessel that circulates blood from the lower-left part of the heart, is the largest blood vessel in the human body.
Designed to mimic the ascending and arch anatomy, Nexus features a pre-shaped catheter that Endospan says is intended to minimise arch manipulation.