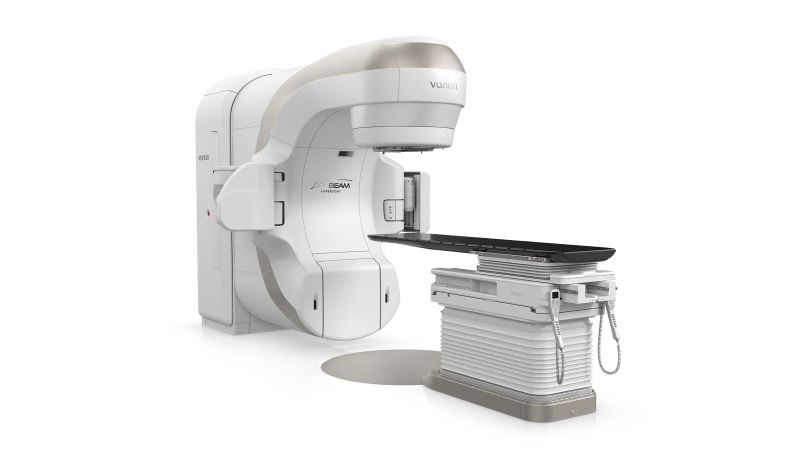
Varian has received US Food and Drug Administration (FDA) clearance for its TrueBeam radiotherapy systems to use low-dose radiation therapy (LDRT) in adults with medically refractory osteoarthritis (OA).
Applying to the Siemens Healthineers subsidiary’s systems including TrueBeam, TrueBeam STx, VitalBeam, and Edge, Varian highlighted that clearance expands its radiotherapy systems’ application beyond cancer care into chronic musculoskeletal conditions.
Research indicates that OA affects around 33 million people in the US, with a rapid upswell in the condition’s incidence in the US and globally due to factors including ageing, obesity, and injury.