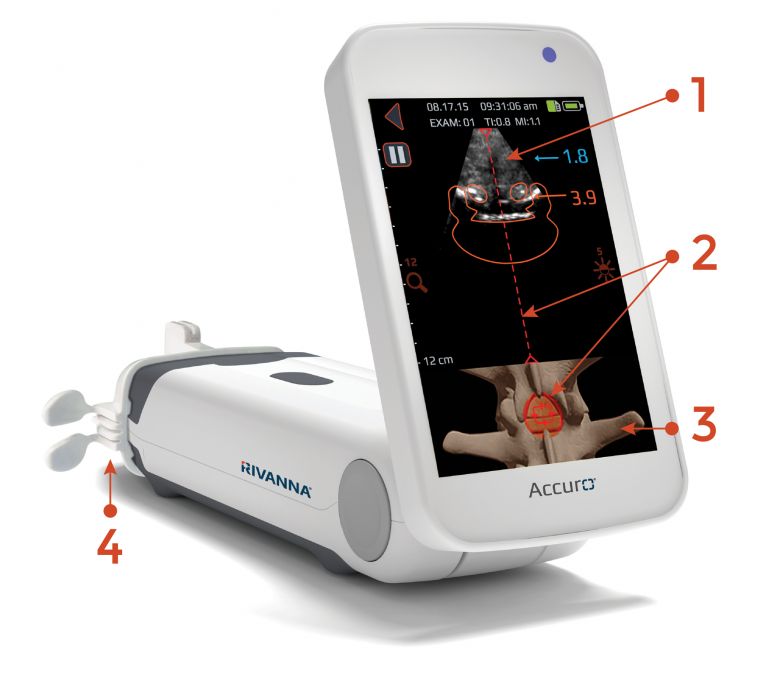
Rivanna’s Accuro XV has secured US Food and Drug Administration (FDA) clearance, setting the stage for the artificial intelligence (AI)-based musculoskeletal imaging system’s commercialisation across hospitals and medical clinics in the US.
Accuro XV uses ultrasound-based bone and 3D soft-tissue imaging technology, along with employing AI-based software to automate the detection and volumetric assessment of skeletal fractures and soft tissue injuries.
Designed for use at the point-of-triage, the system is intended to streamline emergency department workflows, hasten the evaluation of low-severity extremity injuries, and reduce reliance on x-ray referrals, Rivanna stated.