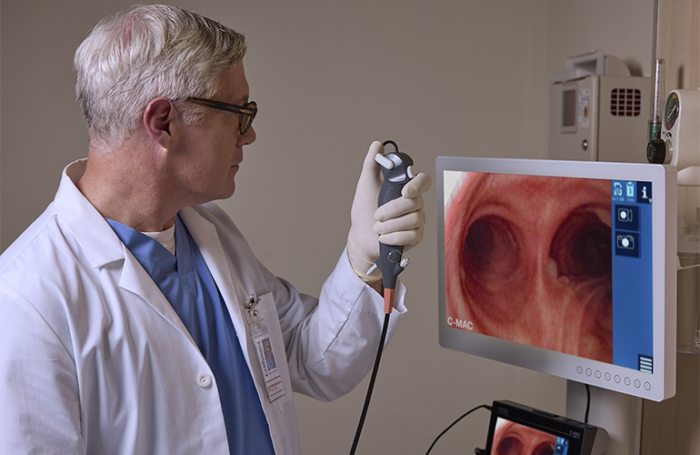
The company says its new device offers a valuable new option for critical care physicians, intensivists, RTs and ICU-based proceduralists. It delivers an airway control solution for high-risk patients, especially those managing heavy secretions or in urgent situations requiring fast airway clearance.
FIVE S 6.5 delivers powerful suction with a 3 mm working channel. During bench testing, it cleared 15 mL of simulated mucus (prepared using egg whites) in less than five seconds.
Karl Storz said it designed the bronchoscope to perform like a reusable scope. It incorporates several user-centric features that prove vital in critical care.