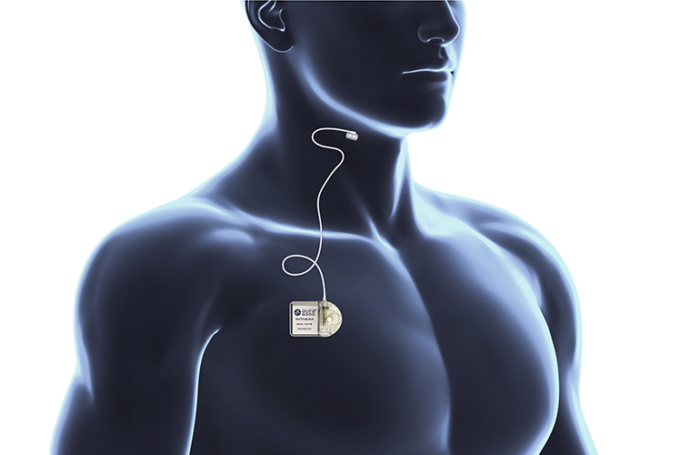
The system utilizes proximal hypoglossal nerve stimulation (p-HGNS), a differentiated neurostimulation modality. It aims to treat obstructive sleep apnea (OSA) in patients with an apnea-hypopnea index (AHI) between 15 and 65 who have failed, do not tolerate, or are ineligible for first-line therapies, such as positive airway pressure (PAP).
London-based LivaNova’s system could rival on-the-market neurostimulators from Inspire Medical and Nyxoah. They all offer an alternative to traditional CPAP sleep respiratory systems. LivaNova submitted the technology to the FDA in May 2025.