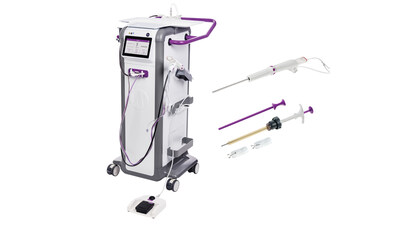
The Mammotome Prima™ MR system features an intuitive in-room design1 that simplifies setup and operation. Clinicians benefit from larger tissue samples2 enabled by stronger vacuum3 and an 8-gauge needle, made possible by 75% less tubing compared to other systems4. The reduced tubing also supports faster setup and cleanup. A touchscreen interface allows convenient adjustments to aperture size and vacuum strength.
FDA clearance of the HydroMARK™ Plus Breast Biopsy Site Marker for exclusive use with the Mammotome™ Prima MR universal targeting set delivers added confidence for MR-guided breast biopsy procedures. Its intuitive integration with the targeting sleeve5 allows consistent and accurate deployment into the biopsy cavity 6. The HydroMARK™ Plus marker incorporates the exclusive hydrogel technology and titanium shapes that provide visibility under both T1 and T2 MRI sequences without excessive bloom5. In addition, the marker is designed to attach to tissue upon deployment and features “wings” designed to mitigate displacement during surgical excision7. Its long-term ultrasound visibility – lasting up to 12 months, even after neoadjuvant chemotherapy – supports reliable pre-surgical localization and continuity from biopsy through surgery8,9.