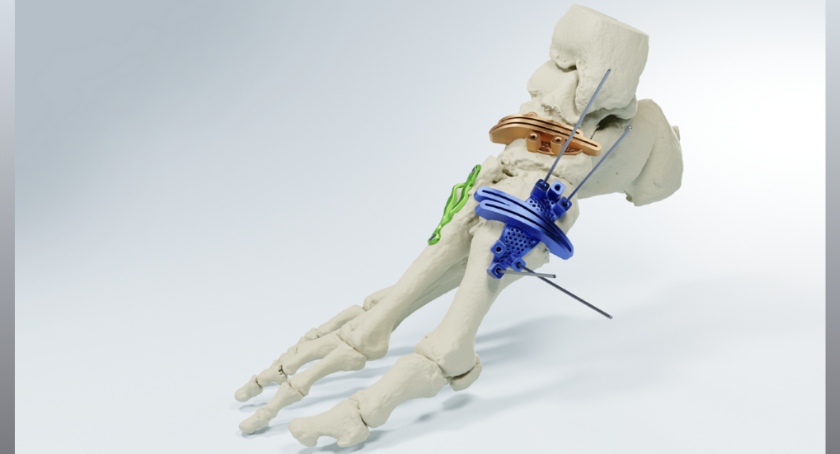
The company recently received 510(k) FDA clearance for its foot and ankle guides and planning system in March 2025. In November 2025 MedCAD received 510(k) FDA clearance for its fixation plates, part of its patient-specific 3D-printed solution available to surgeons as a complete foot and ankle (F&A) system. The newest 510(k) FDA clearance now completes the patent-pending AccuStride® system offering for lower extremities: tibia/fibula, foot and ankle.
“MedCAD’s newly announced, patient-matched surgical guides are unlike anything else available for surgeons who routinely perform complex or revision lower leg region cases,” said Nancy Hairston, CEO and president of MedCAD. “Interest from leading orthopedic specialists eager to use our solutions for multiple pathologies is very promising, and we expect these custom 3-D printed devices to reduce the frequency and duration of surgeries, and to deliver high quality, durable outcomes.”