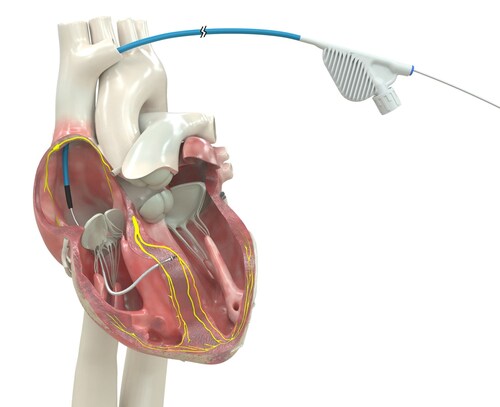
GALWAY, Ireland, March 23, 2026 /PRNewswire/ — Medtronic plc (NYSE: MDT), a global leader in healthcare technology, received U.S. Food and Drug Administration (FDA) approval for an expanded indication for the OmniaSecure™ defibrillation lead. Now approved for placement in the left bundle branch (LBB) area, the lead can be used for conduction system pacing (CSP), which closely mimics the heart’s natural physiology. Additionally, patients in need of cardiac resynchronization may benefit from left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel therapy that combines CSP with left-ventricular pacing to further improve patient outcomes.
The OmniaSecure defibrillation lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. In addition to being the first defibrillation lead approved for placement in the LBB area, the OmniaSecure lead is the smallest diameter defibrillation lead on the market (4.7 French, or 1.66 mm) and the only one approved for adults and adolescent pediatric patients ages 12 and up.