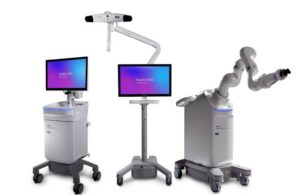
Approval in Europe covers spine and cranial procedures for the planning, navigation and robotics platform. It follows back-to-back FDA nods for Stealth AXiS to kick off 2026, first garnering clearance for spine surgery, then getting the green light for cranial and ENT procedures. Medtronic also has a partnership with GE HealthCare to integrate the platform with intraoperative ultrasound technology.
A spine center in Virginia said it became the first in the world to perform surgery using Stealth AXiS last week.