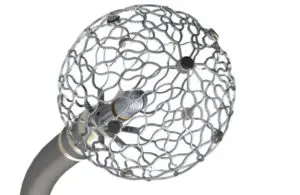
The medtech giant said the FDA granted breakthrough device designation for its Sphere-9 catheter for the treatment of ventricular tachycardia (VT). At Heart Rhythm Society (HRS) 2026 in Chicago, the company also presented new data supporting the use of Sphere-9 for VT, in addition to new findings for the Sphere-360 system.
Medtronic’s Affera mapping and ablation platform makes up a key component of Medtronic’s PulseSelect pulsed field ablation (PFA) system. It became the first such system FDA-cleared for treating AFib in 2024. Medtronic brought in these technologies through its $1 billion acquisition of Affera in August 2022.