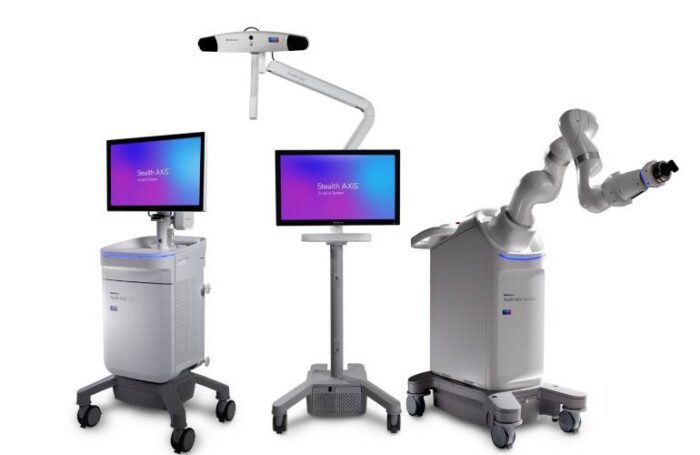
The Stealth AXiS system already held clearance for spine surgery, which it picked up last month. Now, its indications also cover cranial and ear, nose and throat (ENT) procedures.
Stealth AXiS brings planning, navigation and robotics together into a single, intelligent system. Michael Carter, SVP and president of Medtronic Cranial & Spinal Technologies, spoke to MassDevice recently to outline the benefits of the system.