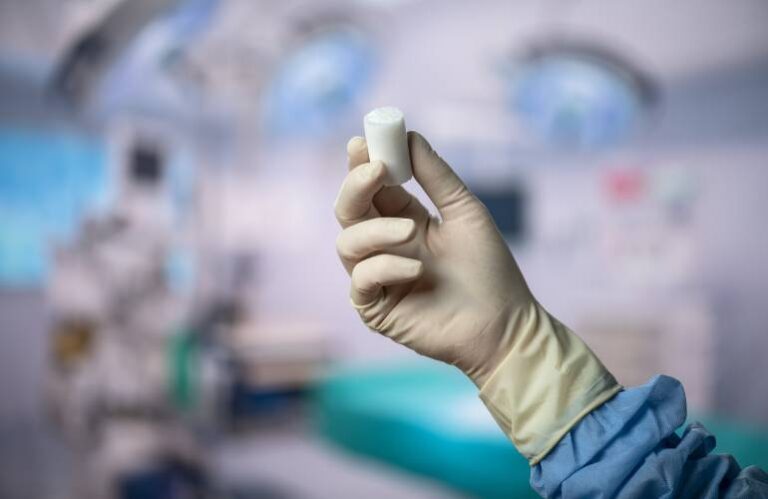
The Bear implant previously won novo approval to treat skeletally mature patients at least 14 years of age with complete ACL tears. Now, the FDA granted 510(k) clearance to expand the indication to include children and adolescents of any age. It also expanded the device to partial ACL tears.
Bear enables a torn ACL to heal and restores the natural function of the knee. The implant restores ACL quality and size similar to the non-injured ACL to provide faster recovery of muscle strength. It delivers better patient satisfaction around returns to sports. By eliminating the need for a graft, BEAR allows for patients to avoid needing a second wound site to heal. This alleviates worries about donor graft quality or risk of disease.
Miach Orthopaedics says the approval offers a new option for young athletes who tear their ACLs each year. It offers a more natural solution to ACL injuries and works with a child’s body as the musculoskeletal system continues to develop.