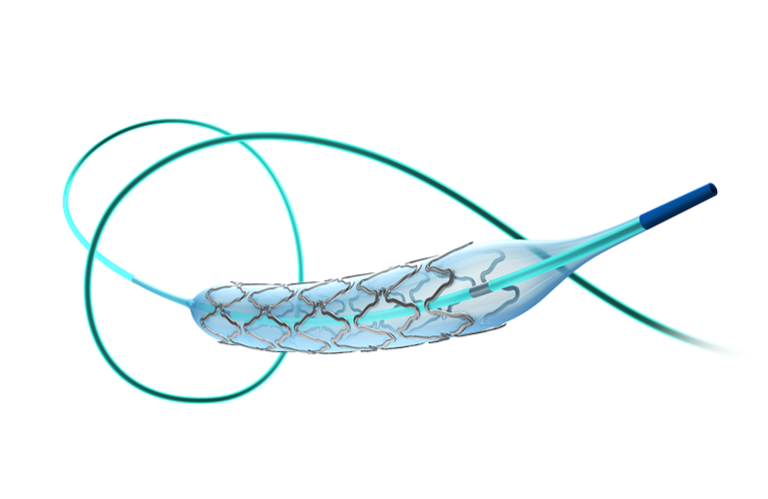
Apollo Dream, an investigational, balloon-expandable, rapid-exchange drug-eluting stent system, is designed to provide vascular scaffolding and restore. cerebral blood flow. It aims to treat intracranial atherosclerotic disease (ICAD), a leading cause of ischemic stroke.
The stent incorporates a proprietary microgroove drug delivery architecture. Sirolimus and a bioabsorbable polymer precisely fit within micron-sized grooves on the abluminal (vessel wall-facing) surface of the stent. The luminal (blood-facing) surface remains in a bare-metal state.
As the polymer degrades, sirolimus goes to the vessel wall. The company said its drug load registers lower than traditional drug-eluting stents.