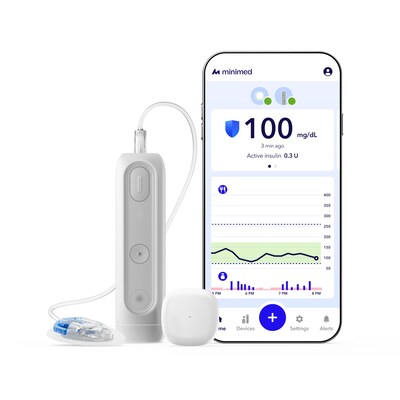
NORTHRIDGE, Calif., March 18, 2026 /PRNewswire/ — MiniMed (Nasdaq: MMED), a global leader in diabetes technology, today announced U.S. Food and Drug Administration (FDA) clearance of MiniMed Flex™, a next-generation discreet, smartphone-controlled insulin pump designed to fit seamlessly into everyday life.
About half the size* of the MiniMed™ 780G pump and roughly the size of two stacked insulin vials, MiniMed Flex™ is the company’s first screenless design created in partnership with people living with diabetes to deliver an intuitive, lifestyle‑friendly way to manage the condition. MiniMed Flex™ is powered by the company’s most advanced SmartGuard™ algorithm with Meal Detection™ technology, which has proven clinical performance that exceeds worldwide Time in Range recommendations.1,2 At commercial launch, MiniMed Flex™ will support the company’s newest sensor portfolio, including Simplera Sync™ sensor and the Instinct sensor, made by Abbott.