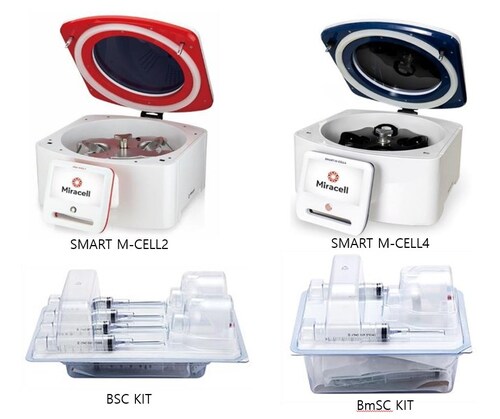
SEOUL, South Korea, April 10, 2026 /PRNewswire/ — Miracell, a leading stem cell technology company, announced that it has obtained U.S. FDA 510(k) clearance for its stem cell extraction system, SMART M-CELL, along with its dedicated kits, the BSC Blood Kit and BMSC Bone Marrow Kit.
The clearance was granted based on demonstrated substantial equivalence to SmartPReP, a globally recognized standard system originally developed by the Institute for Immunological Research (IDI) at Harvard Medical School. This milestone underscores the international credibility of Miracell’s technology in terms of cell quality and performance.