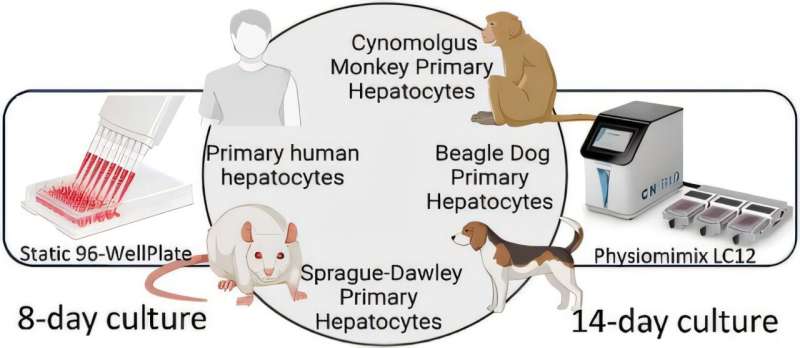
It can be especially challenging to predict how new drugs will affect the liver, the body’s primary organ for processing medications and removing toxins. If a medicine turns out to be harmful to the liver, it can cause a condition known as drug-induced liver injury, which is one of the most common reasons a new drug may never reach patients, even if it may have promising anti-disease properties.
Traditionally, scientists use cells in a dish, followed by tests in animals, to determine the safety of new medications, but the many anatomical and physiological differences between humans and the animals used for safety experiments, especially concerning the liver, make this an often-unreliable process.