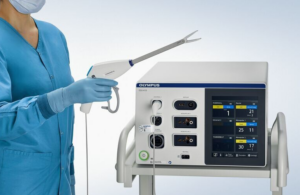
The company adds the device to three 5 mm devices in the Powerseal portfolio. It offers these devices in three jaw configurations, each in three shaft lengths, to support surgeon preference and technique. The portfolio offers dissection, grasping and sealing during open and laparoscopic surgical procedures.
Powerseal’s indications include general surgery, gynecological, colorectal, bariatric, urological, thoracic and vascular.