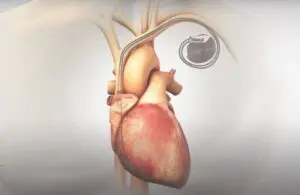
AVIM (atrioventricular interval modulation) uses conduction system pacing to immediately, substantially and persistently reduce blood pressure. The patented bioelectronic therapy comes through a standard dual-chamber pacemaker. New Hope, Pennsylvania–based Orchestra won its first FDA breakthrough device designation for AVIM therapy last year.
Today, the company says the FDA granted a breakthrough nod for AVIM therapy specific to patients with uncontrolled hypertension despite the use of anti-hypertensive medications, and an indication for a pacemaker.