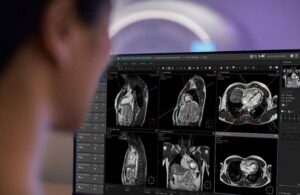
SmartHeart, an AI-powered cardiac magnetic resonance (MR) planning solution, aims to simplify one of the most demanding MRI exams. It automates the planning process in under 30 seconds to make cardiac MR more consistent and accessible.
Cardiac MR exams traditionally require experienced operators. They need to navigate complex scan planning, breath-hold coordination and patient-specific variations. Integrating AI directly into the planning workflow automates these steps in a single click. With this, SmartHeart delivers stable workflows, reduces workload and supports consistent image quality.