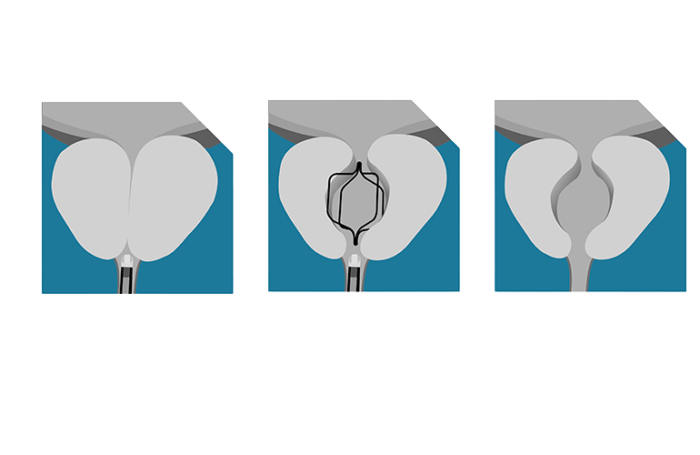
Sunnyvale, California-based Prodeon designed Urocross as a novel, non-permanent implant technology for treating lower urinary tract symptoms (LUTS) associated with benign prostatic hyperplasia (BPH).
It marks the latest device of this kind to pick up an FDA nod related to BPH. ProVerum and Zenflow announced FDA approvals for their implant-based technologies that treat BPH in December 2025. In January of this year, Butterfly Medical also said it completed the final 12-month follow-up for all patients in a study of its prostatic retraction device for BPH. Meanwhile, last week, robotic technology developer Procept launched its BPH-treating system internationally.