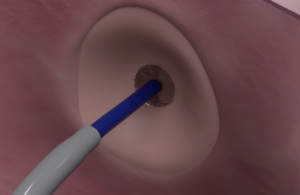
Baltimore-based Protaryx said clearance marks a significant advancement in its mission to redefine safe, efficient and reproducible left heart access. for cardiac interventions. It hopes to enable broader adoption and improved delivery of minimally invasive therapies.
Protaryx designed its technology to support zero-exchange delivery. It features a unique atraumatic design that reduces procedural steps and the need for rewiring. The device incorporates a highly echogenic, extendable atraumatic positioning probe and a standardized RF guidewire. It has compatibility with commercially available electrosurgical generators.