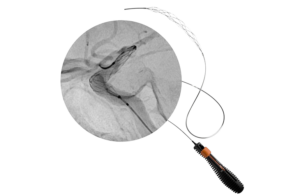
The company also reported the first clinical cases using the device in treating large vessel occlusion (LVO) stroke.
Designed specifically for large clot burden in proximal vessels, Tigertriever 25 combines a 53 mm capture length with proprietary real-time force control. This enables physicians to actively control clot interaction during procedures. The company said this produces consistent performance across a range of clinical scenarios, including long thrombi and stenotic vessels.