Cancer cells survive by repairing damage to their DNA—even damage that would normally be fatal. One of their most important defense systems is homologous recombination, a high-precision repair pathway that fixes broken DNA using key proteins such as RAD51 and CHK1. While therapies such as PARP inhibitors have successfully targeted this vulnerability, many tumors eventually regain their DNA repair ability and become resistant to treatment.
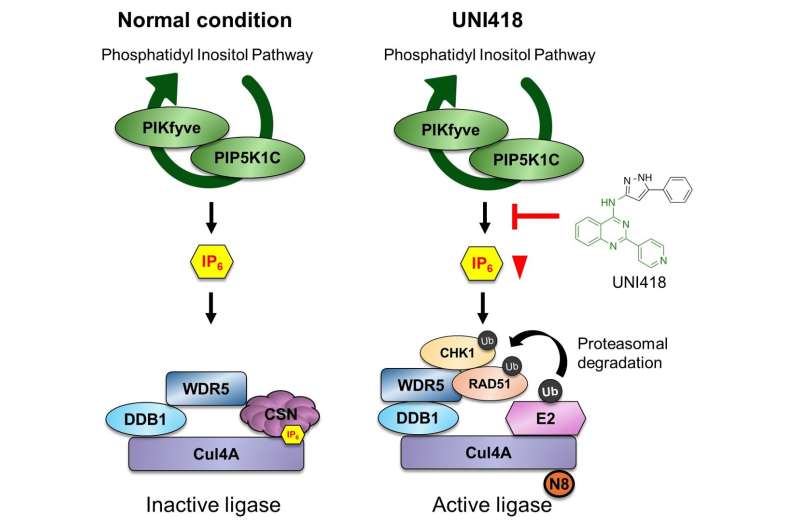
In cells, DNA repair proteins are not static. Their levels are tightly regulated to maintain a balance between repair and genome stability. However, the researchers found that this balance can be deliberately disrupted.
Through a cell-based screening approach designed to identify modulators of replication stress responses, the team discovered a small molecule called UNI418. When applied to cancer cells, UNI418 caused a significant reduction in key DNA repair proteins, including RAD51 and CHK1. As these proteins were depleted, the cells lost their ability to efficiently repair DNA damage.