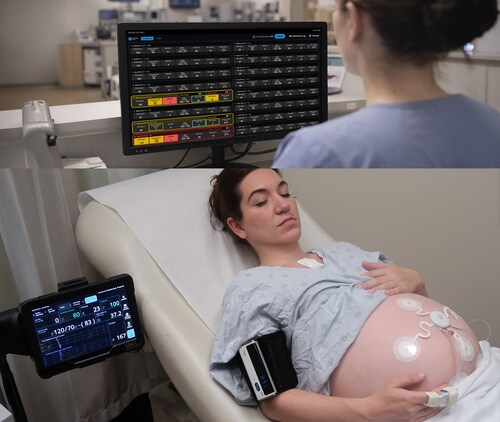
Designed for continuous, simultaneous monitoring of maternal and fetal health, ANNE® Maternal combines advanced wearable sensor technology with integrated clinical decision support — and represents a critical step toward making continuous maternal monitoring a universal standard of care. ANNE® Maternal is a sister product to ANNE® One monitoring platform already FDA-cleared for continuous monitoring of adolescents and adults in the home and hospital setting.
Designed for continuous, simultaneous monitoring of maternal and fetal health, ANNE® Maternal combines advanced wearable sensor technology with integrated clinical decision support — and represents a critical step toward making continuous maternal monitoring a universal standard of care. ANNE® Maternal is a sister product to ANNE® One monitoring platform already FDA-cleared for continuous monitoring of adolescents and adults in the home and hospital setting.