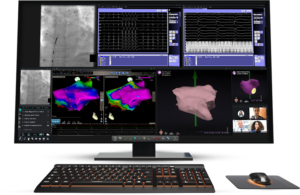
The St. Louis-based developer of surgical robotic technology for minimally invasive endovascular intervention designed its Synchrony platform to digitize and modernize the interventional cath lab. FDA clearance comes about six months after the company submitted Synchrony to the agency. At that time, it also received CE mark for the technology.
Synchrony features a slim, 55” 4K ultra-high-definition display, consolidating the viewing and control of all disparate systems in the lab. It offers an enhanced procedure experience with custom layouts and streamlined workflows. The system also has an intuitive user interface and a decluttered environment.