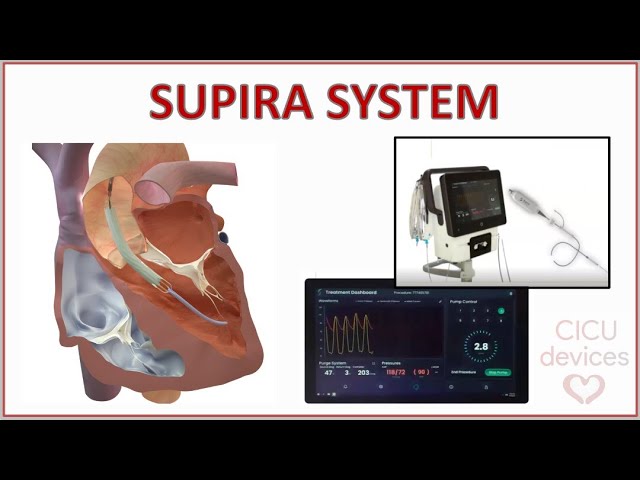
The SUPPORT II pivotal trial evaluates the company’s next-generation pVAD in patients undergoing high-risk percutaneous coronary intervention (HRPCI). Los Gatos, California-based Supira plans to use findings to support a future FDA premarket approval (PMA) submission.
About a year ago, the company raised $120 million to support this trial. It follows an early feasibility study launched the year prior.
Supira said the FDA’s green light for the study marks a “critical step” toward U.S. market entry.