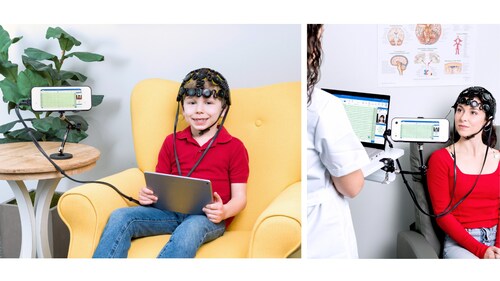
Patients across the U.S. often wait weeks for an EEG appointment, delaying critical answers and care. Clinics are also under pressure, facing shortages of specialized EEG technologists and relying on traditional setups that can be slow and cumbersome. Together, these challenges can significantly delay both patient testing and related follow-up care.
Zeto New Wave™ is designed to change that. It offers a complete, easy-to-use outpatient routine EEG solution, featuring a full head coverage with 21 soft-tip, no mess electrodes placed according to the widely used 10–20 system. Paired with a compact recording device with built-in display and controls, the system also captures synchronized video and audio, along with optional signals such as ECG, EOG, EMG, orientation sensors, external triggers for event related potentials (ERPs), and integrated photic stimulation detection.