
Medtronic wins CE mark for OmniaSecure defibrillation lead
Medtronic (NYSE:MDT) today announced CE mark approval and the first commercial European implants for its OmniaSecure system.

Medtronic (NYSE:MDT) today announced CE mark approval and the first commercial European implants for its OmniaSecure system.
As spring approaches, a new type of cicada is emerging — not an insect, but a new variant of COVID-19.
An acute pulmonary embolism occurs when a blood clot blocks one or more vessels in the lung. In patients at moderate or high risk, the blockage can impair the heart’s ability to pump blood through the lungs to such an extent that it becomes life-threatening. About 15% of those affected die as a result of acute pulmonary embolism. Blood-clot-dissolving medications, known as thrombolytics, can effectively dissolve vascular blockages. However, they increase the risk of severe bleeding, particularly in the brain. For this reason, they are generally used only in the most severe cases.
Scientists led by Botond Roska at the Institute of Molecular and Clinical Ophthalmology Basel (IOB) and collaborators have identified genetic pathways and compounds capable of protecting cone photoreceptors from the degeneration that underlies conditions like age-related macular degeneration.

Over one billion people worldwide are over 60, and the population is projected to more than double by 2050. But as more people live into their 60s, 70s, and 80s, health care systems across the globe may face new challenges as they attempt to manage associated increases in age-related disease.

A new study from the University of Chicago taps an ingredient most often used in the lithium-ion batteries that power our devices to open new avenues in biomedical technology. Lithium plays vital roles in the body, but taking it orally can have unwanted side effects—so a pair of UChicago chemistry labs teamed up to find a way to deliver lithium only to the exact places where it’s needed.
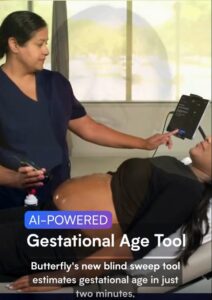
Butterfly’s GA tool within its point-of-care ultrasound (POCUS) system is intended for rollout in regions with socioeconomic barriers to obstetric imaging services.
The algorithm integrates with EHR systems and ECG platforms, operating within the health system without external data transfer.
BOSTON, March 30, 2026 /PRNewswire/ — Haemonetics Corporation (NYSE: HAE), a global medical technology company focused on delivering innovative solutions designed to improve patient outcomes, today announced U.S. Food and Drug Administration (FDA) approval to expand the labeling for the VASCADE MVP® XL venous vascular closure system to include procedures using 10-14F inner diameter (ID) and up to 17F outer diameter (OD) procedural sheaths. With this label expansion, the VASCADE MVP XL system is approved for larger sheaths used in market-leading technologies for pulsed field ablation (PFA) and left atrial appendage closure (LAAC) to treat atrial fibrillation.

New technology designed to provide a single irrigation management system for ureteroscopy, cystoscopy, percutaneous nephrolithotomy (PCNL) and benign prostatic hyperplasia (BPH) procedures