SwiftMR works alongside OEM DL solutions in two ways:
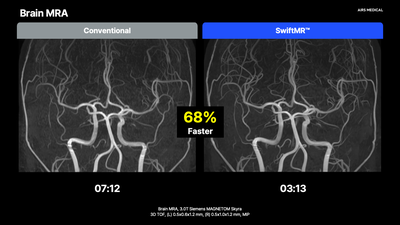
In cases where an OEM DL solution is already installed, SwiftMR works in conjunction to deliver additional scan time reduction. At an imaging center with a GE 3T scanner using AIR Recon DL, SwiftMR further reduced already-accelerated routine brain scans from 15 to 9 minutes.
In cases where an OEM DL solution does not cover a sequence, SwiftMR extends coverage and enables AI-powered image enhancement and scan time reduction across all pulse sequences and all body parts. SwiftMR’s full coverage is FDA cleared and has been in clinical use since 2023.