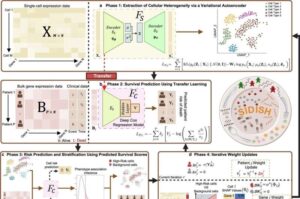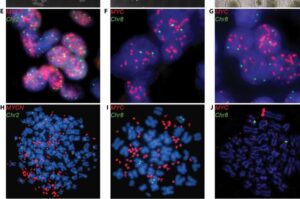
3D brain tumor organoids provide new scientific opportunities for research community
Scientists at St. Jude Children’s Research Hospital have developed patient-derived tumor organoids and tumor organoid xenografts that accurately reflect the biologic underpinnings of embryonal brain tumors.