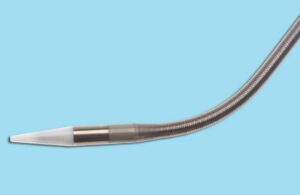
Alleviant Medical wins FDA IDE, breakthrough nods for no-implant heart failure treatment
Alleviant Medical announced today that it received FDA investigational device exemption (IDE) and breakthrough device designation for its atrial shunt.

Alleviant Medical announced today that it received FDA investigational device exemption (IDE) and breakthrough device designation for its atrial shunt.

AccurKardia announced today that it received FDA breakthrough device designation for its ECG-based, AI-powered hyperkalemia detection software.

Insulin delivery technology developer CeQur today announced a significant funding round to support its commercialization efforts.
Francis Medical announced today that it completed an oversubscribed $80 million Series C equity financing.

Fire1 announced today that it completed a $120 million financing round to accelerate the advancement of its heart failure management system.

OXNARD, Calif., Jan. 6, 2025 /PRNewswire/ — PreEvnt, a subsidiary of SCOSCHE Industries, a leading innovator of award-winning consumer technology will proudly unveil isaac* by PreEvnt at the Digital Health Exhibit at CES.

Medtronic (NYSE: MDT)+
today announced CE mark approval for its Harmony transcatheter pulmonary valve (TPV) system
PieX AI announced today that it launched a pendant featuring on-device AI for managing mental health through sensing technology.
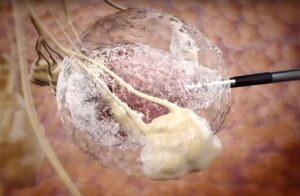
IceCure Medical (Nasdaq:ICCM) announced today that it received an intention to grant notice from the European Patent Office.

Qalo today unveiled its latest silicone smart ring technology for health tracking, called QRNT (pronounced “current”).