
FDA clears new stemless anatomical total shoulder implant from Smith+Nephew
Smith+Nephew (NYSE: SNN)+
announced that it received FDA clearance for its stemless anatomic total shoulder for the Aetos system.

Smith+Nephew (NYSE: SNN)+
announced that it received FDA clearance for its stemless anatomic total shoulder for the Aetos system.

CardioFocus announced today that investigators completed the first series of treatments in a study of its OptiShot pulsed-field ablation system.
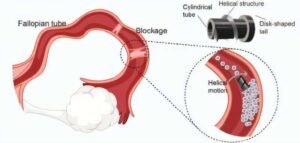
Infertility affects an estimated 186 million people worldwide, with fallopian tube obstruction contributing to 11–67% of female infertility cases. In AIP Advances researchers at the SIAT Magnetic Soft Microrobots Lab have developed an innovative solution using a magnetically driven robotic microscrew to treat fallopian tube blockages.
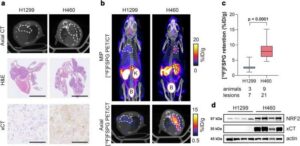
Researchers have used a chemical compound to light up treatment-resistant cancers on imaging scans, in a breakthrough that could help medical professionals better target and treat cancer.

ABBOTT PARK, Ill., Dec. 17, 2024 /PRNewswire/ — Abbott (NYSE: ABT) today announced the successful completion of the world’s first in-human leadless left bundle branch area pacing (LBBAP) procedures using the company’s investigational AVEIR™ Conduction System Pacing (CSP) leadless pacemaker system, as part of a feasibility study.

MannKind today announced six-month results from its Phase 3 INHALE-1 study of Afrezza insulin inhalation powder in children.

DÜSSELDORF, Germany, Dec. 16, 2024 /PRNewswire/ — Gerresheimer, an innovative system and solution provider and a global partner for the pharma, biotech and cosmetics industries, announces that the US Food and Drug Administration (FDA) granted SQ Innovation Tentative Approval for Lasix ONYU for the home treatment of fluid overload in congestive heart failure.

The Osseofit devices are intended to match patients’ shoulder bone anatomy and preserve healthy bone in total shoulder replacement procedures.

HeartBeam (Nasdaq:BEAT) announced today that it received FDA 510(k) clearance for its comprehensive arrhythmia assessment system.

Withings Health Solutions announced today that it received FDA clearance for BPM Pro 2, a first-of-its-kind cellular blood pressure monitor.