
3D-printed brain models could improve medical research and training
University of Missouri researchers are developing new ways to better simulate the complex nature of human brain tissue.

University of Missouri researchers are developing new ways to better simulate the complex nature of human brain tissue.
Stimulating two brain areas, nudging them to collectively fire in the same way, increases a person’s ability to behave altruistically.

Scientists at the University of Maryland have created Smart Underwear, the first wearable device designed to measure human flatulence.

ANDOVER, Mass., Feb. 9, 2026 /PRNewswire/ — TransMedics Group, Inc. (“TransMedics”) (Nasdaq: TMDX), a medical technology company that is transforming organ transplant therapy for patients with end-stage lung, heart, and liver failure, today announced that the U.S. Food and Drug Administration (FDA) has granted full approval of its Investigational Device Exemption (IDE) for the Next-Generation OCS ENHANCE Heart trial.

Researchers detail a machine learning model that dramatically accelerates the manufacturing timeline of monoclonal antibodies.
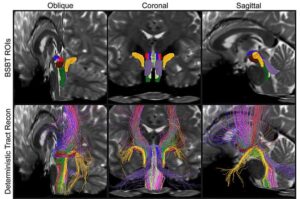
Research reveals distinct patterns of structural changes in patients with Parkinson’s disease, multiple sclerosis, and traumatic brain injury.

Researchers working alongside Australian diagnostics company Nutromics developed a minimally invasive patch that tracks the antibiotic in patients every five minutes.

SEOUL, South Korea, Feb. 6, 2026 /PRNewswire/ — Neurophet (Co-CEOs Jake Junkil Been and Donghyeon Kim), an artificial intelligence (AI) solution company for brain disorders diagnosis and treatment, announced today that its software solution, Neurophet AQUA AD Plus, a comprehensive neuroimaging analysis solution for clinical evaluation related to Alzheimer’s disease, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA).
Eyas Medical Imaging is currently scaling up operations with plans to commercialise the system in the US later this year.
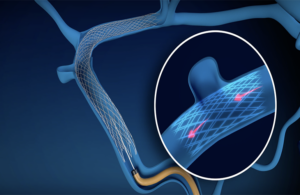
Sonorous Neurovascular received FDA breakthrough device designation for its BosSTENT device intended to treat pulsatile tinnitus, the company announced on Thursday.