
Rethinking ultrasound gel: A natural solid pad for clearer, more comfortable imaging
Ultrasonography is a noninvasive imaging technique used for real-time imaging. This versatile technique is used as a reliable diagnostic tool in various modalities.

Ultrasonography is a noninvasive imaging technique used for real-time imaging. This versatile technique is used as a reliable diagnostic tool in various modalities.
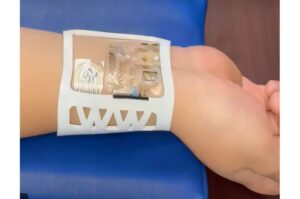
Researchers at the University of California, Irvine have developed a multimodal, bioelectronic wrist-worn device for objective, continuous, real-time monitoring of stress.
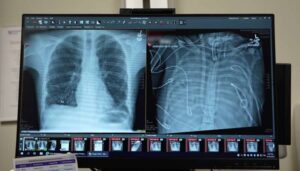
Humans can’t live without lungs, but Ankit Bharat’s patient did for 48 hours.

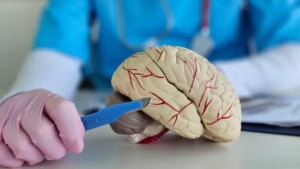
A team led by researchers at Chalmers University of Technology, Sweden, has succeeded in identifying biomarkers for Parkinson’s disease in its earliest stages, before extensive brain damage has occurred. The biological processes leave measurable traces in the blood, but only for a limited period.
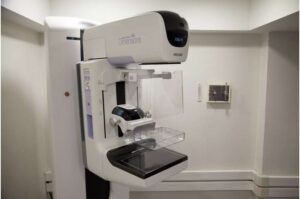
Artificial intelligence (AI)-supported mammography identifies more cancers during screening and reduces the rate of breast cancer diagnosis by 12% in the years following, finds the first randomized controlled trial of its kind. The trial involved over 100,000 Swedish women, and its results are published in The Lancet.
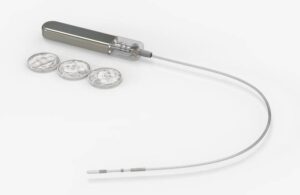
Glucotrack (Nasdaq:GCTK) announced today that it received new U.S. patents for its continuous blood glucose monitoring (CBGM) platform.

From M&A to surgical robotics and user fee negotiations, the medical device industry has a busy year ahead. Check out MedTech Dive’s roundup of the top medtech trends to watch in 2026.
The FDA has authorised up to 15 clinical sites to take part in the study.

Imperative Care announced the launch of the new Zoom 4S thrombectomy catheter and reported the first patient cases with the device.

Pulsed-field ablation, transcatheter heart valves, and intravascular lithotripsy are among the top stories, as they drive growth among the largest cardio device companies.