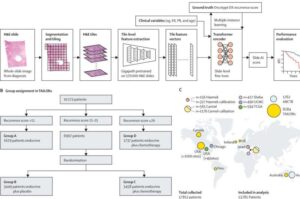
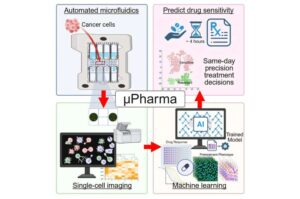
Solving the oxygen problem in cell-based drug delivery
Researchers at Rice University and collaborators at Carnegie Mellon University and Northwestern University have now successfully integrated solutions to several persistent challenges to implantable drug factories into a single device.