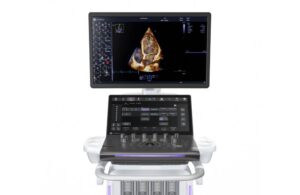
GE HealthCare launches new AI-powered cardiovascular ultrasound system
GE HealthCare (Nasdaq: GEHC)+
today announced the launch of Vivid Pioneer, its newest, most advanced cardiovascular ultrasound system.

GE HealthCare (Nasdaq: GEHC)+
today announced the launch of Vivid Pioneer, its newest, most advanced cardiovascular ultrasound system.
A first-of-its-kind Blood Pressure Treatment Efficacy Calculator built on data from nearly 500 randomized clinical trials in over 100,000 people allows doctors to see how much different medications are likely to lower blood pressure.

PocDoc, a leading UK digital health diagnostics company, has launched what it says is the world’s first smartphone-based test for type 2 diabetes risk including the gold-standard blood biomarker, HbA1c, a landmark step in diabetes prevention according to the company.

Northwestern unveils NIH Baby Toolbox, the newest national standard to assess infants’ cognitive, emotional growth

OSTRAVA, Czech Republic, Aug. 19, 2025 /PRNewswire/ — Stimvia, a medtech company based in the Czech Republic, has successfully completed a clinical study on peroneal electrical transcutaneous neuromodulation (peroneal eTNM®) through its URIS® device to treat Parkinson’s disease symptoms.
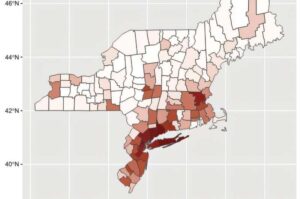
West Nile virus (WNV) has been the dominant cause of mosquito-borne illness in the United States since its introduction into North America in 1999. There are no vaccines nor medications to prevent or treat illness in people, so surveillance, prevention, and control remain the best options to protect the public.

RESEARCH TRIANGLE PARK, N.C., Aug. 19, 2025 /PRNewswire/ — Calidar, Inc., an innovative start-up in precision diagnostic imaging, today announced the successful imaging of the first patient with its 4D Mammography system.

MicroPort NeuroScientific has launched its Numen coil embolization system in Egypt through a partnership with PentaMed.

A smartphone app that delivers real-time, tailored messages may hold the key to helping them quit, according to University of Oklahoma clinical trial results published in JAMA Network Open.

Apple said it was able to bring a redesigned blood oxygen feature to its devices after a recent U.S. customs ruling.