
Insulet wins FDA clearance for Omnipod 5 algorithm enhancements
Insulet (Nasdaq: PODD)+
announced today that it received FDA 510(k) clearance for new enhancements to its Omnipod 5 system.

Insulet (Nasdaq: PODD)+
announced today that it received FDA 510(k) clearance for new enhancements to its Omnipod 5 system.
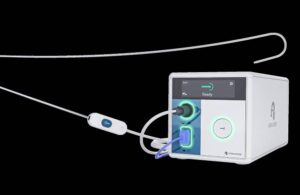
Atraverse Medical announced today that it received FDA clearance for its fully integrated Hotwire transseptal access system.

Boston Scientific (NYSE: BSX)+
announced recently that it received CE mark for its Farapoint pulsed field ablation (PFA) catheter.
The trial will enroll patients who are undergoing nerve management procedures for severe neuropathic pain.

MED-EL’s cochlear implants are now FDA-approved in children seven months and older with bilateral sensorineural hearing loss (SNHL).

The approval is a major endorsement of Biomoneta’s scientific breakthrough and an important inflection point for Beyond Next Ventures India (BNV India)

Avance is an acellular nerve scaffold for the treatment of adult and pediatric patients aged 1 month or older with sensory, mixed, and motor peripheral nerve discontinuities
SURREY, BC, Dec. 4, 2025 /PRNewswire/ – mlHealth 360 today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance (K250694) for Scaida BrainCT-ICH, an AI-powered triage solution designed to address critical bottlenecks in acute care radiology.

MIDDLETON, Wis., Dec. 3, 2025 /PRNewswire/ — Natus Medical Incorporated announced the electrographic status epilepticus diagnosis capability of its BrainWatch point-of-care EEG solution, featuring integrated Persyst analysis software which has received 510(k) clearance from the U.S. Food and Drug Administration.

BEIJING, Dec. 3, 2025 /PRNewswire/ — Wingderm® announces that its Renuva 1550nm non-ablative fractional laser system has obtained MDR certification in the European Union, marking a significant regulatory milestone for the device within the EU market.