Medical Microinstruments wins FDA IDE for robotic microsurgery for Alzheimer’s
Medical Microinstruments (MMI) announced today that it received FDA investigational device exemption (IDE) to study its microsurgery platform

Medical Microinstruments (MMI) announced today that it received FDA investigational device exemption (IDE) to study its microsurgery platform

The panels employ PCR techniques to test for numerous gastrointestinal bacterial pathogens using one stool swab.
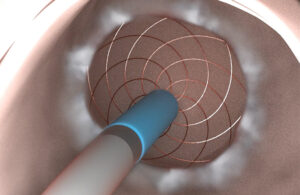
Aqua Medical has received an investigational device exemption (IDE) from the FDA for its ablation device intended to treat diabetes, the company told MassDevice ahead of an official announcement later today.

SAN DIEGO, Nov. 3, 2025 /PRNewswire/ — The U.S. Food and Drug Administration (“FDA”) has granted QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a global leader of in vitro diagnostics, 510(k) clearance for the VITROS hs Troponin I Reagent Pack (the “VITROS hs Troponin I Assay”). The assay is intended for the quantitative measurement of cardiac troponin I (cTnI) in human plasma (heparin) to aid in the diagnosis of myocardial infarction (MI).
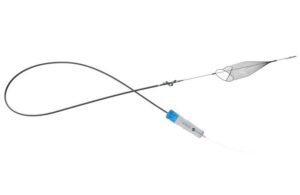
InterVene’s Recana is claimed to be the first fully integrated catheter system to address venous in-stent restenosis (ISR).
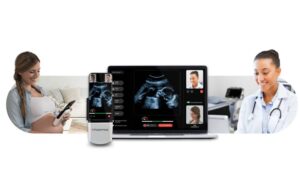
Pulsenmore announced today that it received FDA de novo clearance for its home-use prenatal ultrasound platform.

Wandercraft announced today that the FDA expanded the indications for its flagship Atalante X rehabilitation device.
The clearance from the US regulator broadens the company’s diagnostic imaging offerings.
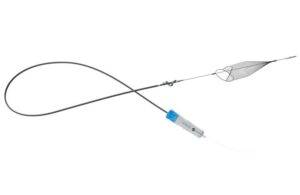
InterVene announced today that it received FDA 510(k) clearance for its Recana thrombectomy catheter system.
Studies showed that the assay demonstrated a 94.90% sensitivity in polymerase chain reaction-confirmed positive samples.