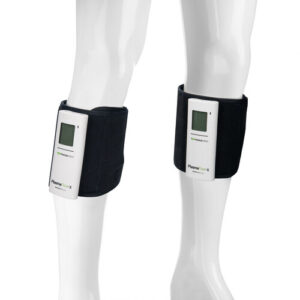
ManaMed® Secures FDA 510(k) Clearance for PlasmaFlow® X — a Breakthrough, Compact Sequential Compression System with All-Day Battery
DENTON, Texas, Oct. 20, 2025 /PRNewswire/ — ManaMed today announced FDA 510(k) clearance for PlasmaFlow® X, the next-generation, tubeless sequential compression system that’s redefining recovery. Cleared as a Class II device, PlasmaFlow X is more compact, smarter, and longer-lasting—delivering powerful therapy in a sleek, portable package.