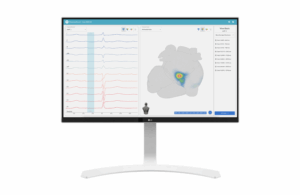
Vektor Medical wins CE mark for AI-based arrhythmia mapping system
Vektor Medical today announced it received CE mark approval for its vMap artificial intelligence-powered arrhythmia mapping system.

Vektor Medical today announced it received CE mark approval for its vMap artificial intelligence-powered arrhythmia mapping system.

GROVE CITY, Ohio, Oct. 6, 2025 /PRNewswire/ — Tosoh Bioscience, Inc., a market leader in clinical diagnostics is proud to announce that it has received US FDA 510(k) clearance for its next-generation system for HbA1c testing – the Tosoh Automated Glycohemoglobin Analyzer HLC®723-GR01 (hereafter “GR01”). The GR01 is intended for monitoring the long-term blood glucose control of individuals with diabetes, as an aid in the diagnosis of diabetes and to help identify those who may be at risk of developing the disease.

GlucoSet announced today on social media that it received FDA breakthrough device designation for its glucose monitoring technology.
IceCure Medical (Nasdaq:ICCM) announced today that the FDA granted marketing authorization for its ProSense cryoablation system for breast cancer.
Hologic’s FDA clearance for its Panther Fusion GI Bacterial and Expanded Bacterial Assays represents the company’s entry into the gastroenteritis test market.
Distalmotion announced today that it received FDA 510(k) clearance for the use of its Dexter robotic surgery system in hysterectomy.
Medtronic (NYSE: MDT)+
announced today that it received CE mark approval for its Penditure LAA exclusion system.
NEW YORK, Sept. 30, 2025 /PRNewswire/ — Aidoc, the global leader in clinical AI, today announced the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the company’s novel multi-triage solution that flags a wide array of life-threatening, time-sensitive medical conditions, all within a single workflow. Built on CARE™, the first clinical-grade foundation model in healthcare with FDA cleared solutions, and deployed through Aidoc’s aiOS™ platform, the solution is designed to help care teams attend to high–risk cases faster and more consistently across the health system.

SAN FRANCISCO, Sept. 30, 2025 /PRNewswire/ — Avive Solutions, Inc. is proud to announce a significant advancement in public safety and emergency preparedness: the smallest, lightest AED on the market, the Avive Connect AED®, is safe and effective for use on aircrafts. Following rigorous testing and evaluation, this clearance confirms that the device is safe and effective for use on aircraft, enabling a new standard of emergency response at 30,000 feet.

Zenflow announced today that it received FDA 510(k) clearance for its Spring scope and camera control unit (CCU).