
Zeta Surgical wins FDA clearance for TMS navigation system
Zeta Surgical this week announced it received FDA 510(k) clearance for its Zeta TMS Navigation System

Zeta Surgical this week announced it received FDA 510(k) clearance for its Zeta TMS Navigation System
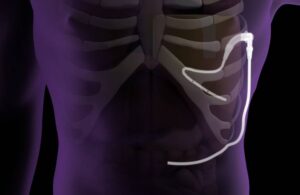
Pleural Dynamics announced today that it received a new CMS code for its ACES (automatic continuous effusion shunt) device.

Medtronic (NYSE: MDT)+
announced today that it received new labeling approval from the FDA for its Endurant stent graft system.
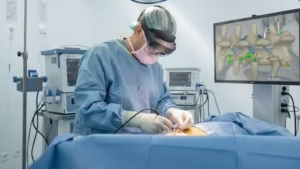
OrthoIndy spine surgeon Dr Greg Poulter performed the inaugural SyncAR Spine case.

The xStep device offers painless, non-invasive electrical stimulation to the spinal cord to treat paralysis.
An Abbott (NYSE: ABT)+
official posted on social media to announce FDA breakthrough device designation for a new ablation catheter.
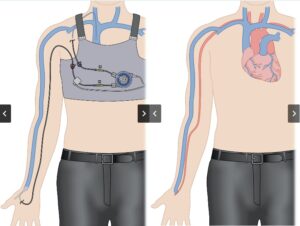
ST. LOUIS, Oct. 9, 2025 /PRNewswire/ — Amplifi Vascular, developer of the first-of-its-kind Vein Dilation System designed to increase eligibility, improve creation and early maturation of arteriovenous fistulas (AVFs) for hemodialysis, today announced three major milestones: positive first-in-human (FIH) study results, the close of a $6.9 million Series A financing, and U.S. Food and Drug Administration (FDA) approval of its Investigational Device Exemption (IDE).
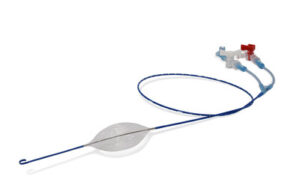
BOERNE, Texas, Oct. 8, 2025 /PRNewswire/ — Building once again on their first mover position as a leader in endovascular hemorrhage control and resuscitation, Prytime Medical Devices, Inc. (Prytime) this week announced a new FDA 510(k) clearance for their flagship pREBOA-PRO™ Catheter.
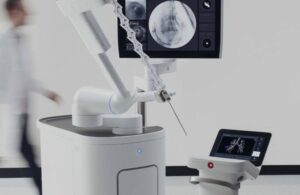
ntuitive Surgical (Nasdaq: ISRG)+
announced today that it received FDA clearance for new software advancements for its Ion endoluminal robot.

SALT LAKE CITY, Oct. 7, 2025 /PRNewswire/ — Xenocor, Inc., a leader in single-use surgical visualization, proudly announces the issuance of U.S. Patent No. 12,429,685 B2 for its innovative medical borescope and tip assembly technology. This milestone reinforces Xenocor’s commitment to advancing safe, efficient, and accessible minimally invasive procedures through disposable and digitally optimized laparoscopic tools.