
Outlook Surgical wins FDA clearance for hybrid endoscope system
Outlook Surgical this week announced it received FDA 510(k) clearance for its Inova 1 Towerless Endoscope System.

Outlook Surgical this week announced it received FDA 510(k) clearance for its Inova 1 Towerless Endoscope System.
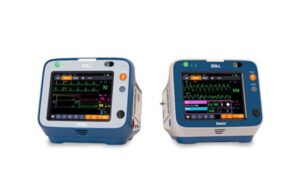
Zoll announced today that it received FDA premarket approval (PMA) for its Zenix professional monitor/defibrillator.

Abbott (NYSE: ABT)+
said Health Canada authorized its Esprit BTK Everolimus Eluting Resorbable Scaffold System, a dissolving stent for patients with chronic limb-threatening ischemia (CLTI) below the knee.

Tandem Diabetes Care (Nasdaq:TNDM) announced today that it received approval to deliver Lyumjev ultra-rapid acting insulin with its t:slim X2 pump.

iTaperloc Complete and iG7 Hip System with Iodine Technology inhibits bacterial adhesion and prevents biofilm formation on the implant surface to help address the issue of Periprosthetic Joint Infection after total joint replacement

Investigators saw a 71% reduction in myopia progression in children who used the lenses for two years.

AngioSafe announced it has received FDA 510(k) clearance and CE marking for its Santreva-ATK endovascular revascularization catheter.
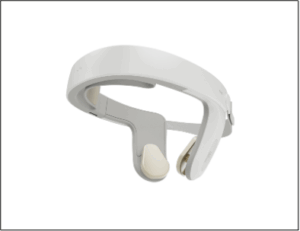
The newly issued patent strengthens Nexalin’s expanding intellectual property estate, supporting the Company’s DIFS platform and ongoing clinical and regulatory initiatives

The updated algorithm shows a 21% improvement in plaque detection, compared to the original version.

Biolinq announced today that it received FDA de novo clearance for its lead product, the Biolinq Shine wearable biosensor.