
Thermo Fisher’s NGS-Based Oncomine Dx Target Test Gains FDA Approval
FDA clears assay as companion diagnostic for ZEGFROVY™ and broad tumor profiling, advancing precision oncology diagnostics.

FDA clears assay as companion diagnostic for ZEGFROVY™ and broad tumor profiling, advancing precision oncology diagnostics.

Dual AI-Powered Imaging Software Set to Enhance Diagnostic Confidence and Workflow Efficiency in MRI
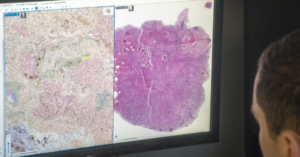
PathAI, a global leader in artificial intelligence (AI) and digital pathology solutions, has announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for AISight Dx—its digital pathology image management system—for use in primary diagnosis in clinical settings.
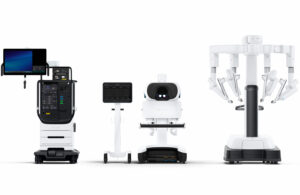
Intuitive Surgical (Nasdaq: ISRG)+
announced today that it received CE mark for its da Vinci 5 surgical robotic system.

Fasikl announced that it received FDA 510(k) clearance for its first-of-its-kind NeuroAI wristband for essential tremor.

PARIS and SAN FRANCISCO, July 2, 2025 /PRNewswire/ — Moon Surgical, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) for two major advancements to its Maestro System: connectivity of the platform to power the Maestro Insights product and a Predetermined Change Control Plan (PCCP) to evolve its AI-powered ScoPilot product.
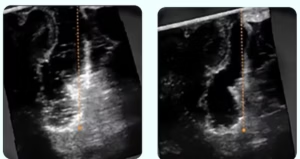
OSLO, Norway, July 1, 2025 /PRNewswire/ — SonoClear AS today announced that the Center for Devices and Radiological Health (CDRH) of the U.S. Food and Drug Administration (FDA) has designated the SonoClear® System as a Breakthrough Device for use in intracranial ultrasound procedures.

Regulatory Approval Paves the Way for U.S. Market Launch of Innovative Therapy Targeting Chronic Pain and Neurological Disorders
BOSTON, June 30, 2025 /PRNewswire/ — TOBY, Inc., a biotech startup advancing non-invasive cancer detection through urine-based volatilomics and Artificial Intelligence, announced today the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its TOBY Test for Bladder Cancer.
New treatment option offers continuous symptom control for patients with advanced Parkinson’s disease, enhancing quality of life through minimally invasive drug delivery