
Element Sciences earns FDA premarket approval for wearable defibrillator patch
Element Science announced today that it received FDA pre-market approval for its Jewel Patch wearable cardioverter defibrillator (WCD).

Element Science announced today that it received FDA pre-market approval for its Jewel Patch wearable cardioverter defibrillator (WCD).

Neuralink said today on social media that it received FDA breakthrough device designation for treating individuals with severe speech impairment.
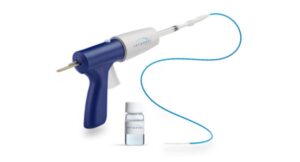
Teleflex (NYSE: TFX) announced today that it received FDA 510(k) clearance for expanded indications of its QuikClot Control+ hemostatic device.
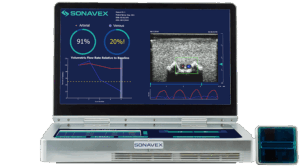
Sonavex announced that it received FDA 510(k) clearance for its AI-enabled EchoMap device with 3D ultrasound capabilities.
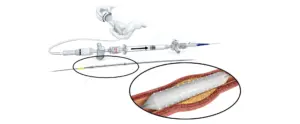
Orchestra BioMed (Nasdaq:OBIO) announced today that it received FDA investigational device exemption (IDE) for its Virtue drug-coated balloon.

William Blair analyst Brandon Vazquez said 2025 could be the first year market leader Intuitive Surgical will face meaningful U.S. competition.

Medtronic (NYSE: MDT) announced that it received FDA approval for its OmniaSecure defibrillation lead for right ventricle placement.
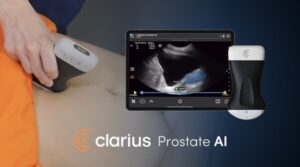
Clarius is the first to offer an FDA-cleared AI-powered prostate measurement tool on a handheld ultrasound scanner—empowering primary care and urology physicians to assess patients on the spot without waiting for specialist referrals

LEM Surgical announced that it received FDA 510(k) clearance for its Dynamis robotic surgical system for hard tissue surgery.
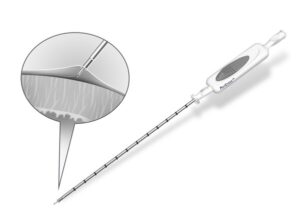
ENGLEWOOD, Colo., April 24, 2025 /PRNewswire/ — CIRCA Scientific is pleased to announce that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the PeriCross™ Epicardial Access Kit (formerly Rook®), a purpose-built solution designed to enable controlled and efficient access to the pericardial space via a subxiphoid approach.