
Zynex submits laser pulse oximeter to FDA
Zynex (Nasdaq:NYXI) announced today that it submitted its NiCO non-invasive co-oximeter device for FDA 510(k) clearance.

Zynex (Nasdaq:NYXI) announced today that it submitted its NiCO non-invasive co-oximeter device for FDA 510(k) clearance.

Milestone Approvals Enable European and UK Market Access for AI-Based Cardiac Arrest Prediction Tool

Women can now collect their own sample from the privacy of home, no speculum required, and mail it to a certified lab to be tested on the same test as the doctor’s office, with the same accuracy.
Final approval clears the way for Zydus to market Glatiramer Acetate, expanding access to a key treatment for multiple sclerosis patients in the U.S.
LivaNova (Nasdaq: LIVN)+
today announced 12-month data from its OSPREY study supporting an FDA submission of the aura6000 system.
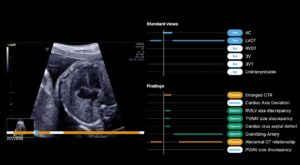
BrightHeart earned its second FDA 510(k) clearance for updates to its BrightHeart platform, an AI-powered digital screening tool for congenital heart defects (CHDs).
GE HealthCare (Nasdaq: GEHC) announced today that it received FDA 510(k) clearance for its Aurora nuclear medicine system and Clarify DL

FaceHeart Vitals™ is a video-based, contactless vital sign measurement SDK that can be integrated into devices such as smartphones seamlessly, providing heart rate, respiration rate, blood pressure and other vital sign readings in 50 seconds.

Edwards Lifesciences (NYSE: EW) announced today that it received FDA approval for its Sapien 3 TAVR for severe aortic stenosis (AS) without symptoms.

Intuitive (NASDAQ: ISRG)+
announced today that it received FDA clearance for a new indication for its da Vinci single port (SP) surgical robot.