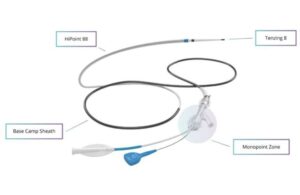
AnX Robotica wins FDA nod for next-gen gastrointestinal motility capsule
The company said in a news release that the latest clearance marks a significant step forward in gastrointestinal (GI) motility monitoring. It offers clinicians an advanced, non-invasive tool for assessing whole-gut transit times with unmatched accuracy and patient comfort.