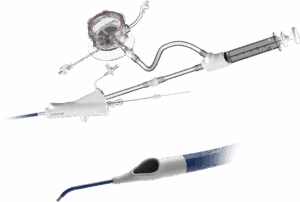
FDA expands clearance for Inquis Medical’s Aventus thrombectomy system
Inquis Medical announced today that the FDA granted expanded 510(k) clearance for its Aventus thrombectomy system.

Inquis Medical announced today that the FDA granted expanded 510(k) clearance for its Aventus thrombectomy system.

GI Windows Surgical announced today that it received FDA 510(k) clearance for its Flexagon self-forming magnet technology.
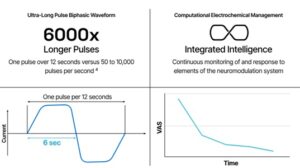
Presidio Medical announced today that it received FDA investigational device exemption (IDE) for its ultra-low frequency (ULF) neuromodulation platform.

Neurent Medical announced that it received FDA 510(k) clearance for its next-generation Neuromark system for chronic rhinitis.

Accelus today announced it received FDA 510(k) clearance for its FlareHawk Interbody Fusion System to be safely scanned under certain MRI conditions

First-in-class gene therapy moves toward clinical trials to address a life-threatening genetic heart condition with high unmet need
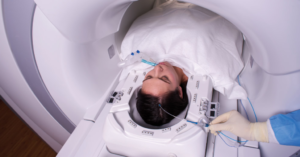
Medtronic has announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Visualase V2 MRI-Guided Laser Ablation System. This milestone brings important capital system enhancements to the Visualase platform, which provides a minimally invasive surgical option for patients with focal epilepsy, brain tumors, and radiation necrosis, which impacts over 1 million people total worldwide.

IceCure has 20+ patents in the U.S. and the Company anticipates further market traction upon FDA’s marketing authorization decision in early-stage breast cancer

HOUSTON, June 9, 2025 /PRNewswire/ — TYBR Health, a medical device and regenerative medicine company, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for the B3 GEL™ System. B3 GEL™ is a bioresorbable, flowable gel barrier designed to protect healing tissue planes and preserve mobility following surgeries involving tendons, ligaments, and skeletal muscle.

Regulatory Milestone Validates JBA AI’s Clinical-Grade Software for Advanced Diagnostic Support