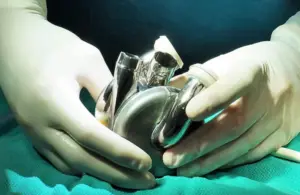
BiVacor wins FDA breakthrough nod for Total Artificial Heart
BiVacor announced today that it received FDA breakthrough device designation for its Total Artificial Heart (TAH) system.

BiVacor announced today that it received FDA breakthrough device designation for its Total Artificial Heart (TAH) system.
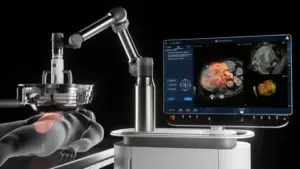
Non-Invasive Histotripsy Technology Authorized Under Unmet Clinical Needs Pathway for Liver Tumor Treatment

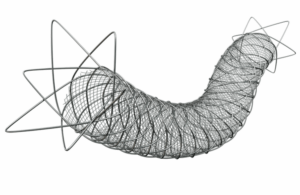
First-of-its-Kind Retrievable Stent Gains Approval for Treating Challenging Below-the-Knee Peripheral Artery Disease
Terumo Interventional Systems has announced the early commercial availability of its FDA-approved Roadsaver Carotid Stent System.
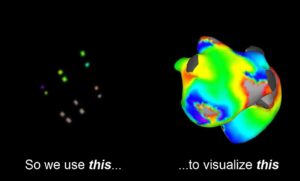
CoreMap announced that it received FDA investigational device exemption (IDE) to extend its electrophysiology (EP) mapping trial to the U.S.
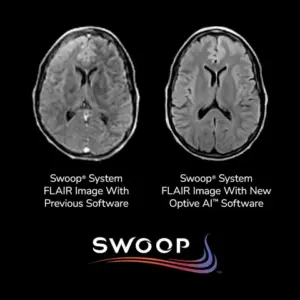
New AI-driven software delivers unprecedented image quality improvements, enhancing diagnostic accuracy and expanding the capabilities of portable MRI technology.

SAN FRANCISCO, May 29, 2025 /PRNewswire/ — Darmiyan, Inc., a leading brain health technology company, today announced that the U.S. Food and Drug Administration (FDA) has approved the company’s post-market surveillance study for BrainSee, its groundbreaking clinical prognostic tool for Alzheimer’s disease. This 7-year study aims to further validate the safety and effectiveness of BrainSee in a socio-demographically diverse patient population across the United States. The insights gained will also support pharmaceutical companies and contract research organizations (CROs) in identifying optimal candidates for next-generation Alzheimer’s clinical trials—advancing both precision and equity in patient selection.

Distalmotion announced today that it received FDA 510(k) clearance for the use of its Dexter surgical robot in adult cholecystectomy (gallbladder removal).
Abbott said the Tendyne system addresses a significant unmet need, providing an option for patients ineligible for open heart surgery and whose valves cannot be successfully repaired with MitraClip.
Medtronic (NYSE: MDT)+
announced today that the FDA accepted its new Nellcor technology into its STeP program.