
Zeiss wins FDA nod for robotic-assisted radiation tech
Zeiss Medical Technology announced today that it received FDA 510(k) clearance for the Intrabeam 700 platform.

Zeiss Medical Technology announced today that it received FDA 510(k) clearance for the Intrabeam 700 platform.

New Approval Expands Use of Porous Biologic Scaffold for Intervertebral Disc Space Fusion Procedures

HighLife announced that it received FDA breakthrough device designation for its trans-septal mitral valve replacement (TMVR) solution.
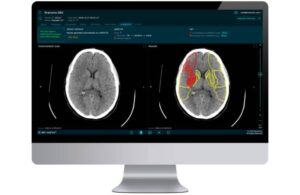
Brainomix announced today that it received FDA clearance for a new feature within its 360 Stroke solution for AI-powered stroke imaging.

Proprio announced today that it received its second FDA 510(k) clearance for its Paradigm AI guidance platform for surgery.

CardioVia announced today that it received FDA clearance for its minimally invasive ViaOne epicardial access system.

Advanced Dosimetry Software Expands Capabilities for Personalized Radiopharmaceutical Therapies

Cutting-Edge Technology Enhances Real-Time Respiratory Tracking for Better Patient Care
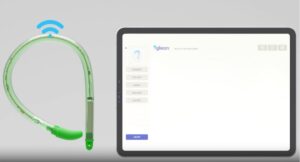
Bright Uro announced today that it received FDA 510(k) clearance for its Glean urodynamic analyzer system.
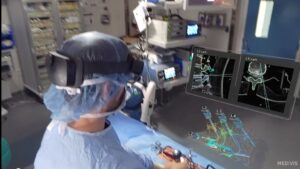
NEW YORK, April 2, 2025 /PRNewswire/ — Medivis Inc., a pioneer in surgical intelligence, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its groundbreaking Spine Navigation platform. This milestone marks a significant advancement in surgical technology, utilizing AR and AI to empower surgeons with holographic navigation across open and minimally invasive spine procedures. Alongside the FDA clearance, Medivis is announcing the commercial launch of Spine Navigation, making it available to hospitals and ambulatory surgical centers nationwide.